Diatomic molecule: Difference between revisions
en>Bibcode Bot m Adding 0 arxiv eprint(s), 1 bibcode(s) and 0 doi(s). Did it miss something? Report bugs, errors, and suggestions at User talk:Bibcode Bot |
en>CorniceBowlSkier mNo edit summary |
||
| Line 1: | Line 1: | ||
{{Use dmy dates|date=July 2013}} | |||
[[Image:Nitrogen-3D-vdW.png|thumb|A [[space-filling model]] of the diatomic molecule dinitrogen, N<sub>2</sub>]] | |||
'''Diatomic molecules''' are [[molecule]]s composed of only two [[atom]]s, of either the same or different [[chemical element]]s. The prefix di- is of Greek origin, meaning "two". If a diatomic molecule consists of two atoms of the same element, such as [[hydrogen]] (H<sub>2</sub>) or [[oxygen]] (O<sub>2</sub>), then it is said to be [[Homonuclear molecule|homonuclear]]. Otherwise, if a diatomic molecule consists of two different atoms, such as [[Carbon_monoxide|carbon monoxide]] (CO) or [[Boron_monoxide|boron monoxide]] (BO), the molecule is said to [[Heteronuclear molecule|heteronuclear]]. | |||
==Homonuclear molecules== | |||
[[File:Diatomic molecules periodic table.svg|thumb|300px|A [[periodic table]] showing the elements that exist as [[homonuclear molecule|homonuclear]] diatomic molecules under typical laboratory conditions.]] | |||
The only [[chemical elements]] which are stable two atom [[homonuclear]] [[molecules]] at [[standard temperature and pressure]] (STP), are [[hydrogen]] (H<sub>2</sub>), [[nitrogen]] (N<sub>2</sub>) and [[oxygen]] (O<sub>2</sub>), plus the [[halogens]] [[fluorine]] (F<sub>2</sub>) and [[chlorine]] (Cl<sub>2</sub>). Those diatomic elements that are gaseous at STP, when grouped together with the [[monatomic]] [[noble gases]], such as [[argon]], are called "elemental gases" or "molecular gases" to distinguish them from molecules that are also [[chemical compounds]]. | |||
The [[noble gases]] do not form diatomic molecules: this can be explained using [[molecular orbital theory]]. | |||
The halogens [[bromine]] (Br<sub>2</sub>) and [[iodine]] (I<sub>2</sub>) can also form diatomic gas at slightly elevated temperatures.<ref name=Chemistry>{{cite book|title=Chemistry|authors=Whitten, Kenneth W.; Davis, Raymond E.; Peck, M. Larry; Stanley, George G.|year=2010|publisher=Brooks/Cole, Cengage Learning|pages=337–338|url=http://books.google.ca/books?id=6Zwu9-qT0qQC&pg=PA337#v=onepage&q&f=false|edition=9th}}</ref><!--Please do not add astatine; see [[Astatine#General characteristics]].--> <br /> | |||
Other elements that can form two atom molecules are [[diphosphorus|phosphorus]] (P<sub>2</sub>) and [[disulfur|sulfur]] (S<sub>2</sub>) although neither of these molecules are stable in atmospheric conditions. | |||
==Heteronuclear molecules== | |||
All other diatomic molecules are [[chemical compounds]] of two different elements, for example, [[nitric oxide]] (NO). Many different elements combine to form heteronuclear diatomic molecules, and this phenomenon, in general, depends on temperature and pressure. Many [[chemical compound]]s form diatomic molecules when evaporated. | |||
==Occurrence== | |||
Hundreds of diatomic molecules have been characterized<ref>{{Cite book| author = Huber, K. P. and Herzberg, G. | title = Molecular Spectra and Molecular Structure IV. Constants of Diatomic Molecules | publisher = New York: Van Nostrand: Reinhold | year = 1979 | id= }}</ref> in the terrestrial environment, laboratory, and [[List of molecules in interstellar space|interstellar medium]]. About 99% of the [[Earth's atmosphere]] is composed of two diatomic molecules: oxygen (21%) and nitrogen (78%). The natural abundance of [[hydrogen|hydrogen (H<sub>2</sub>)]] in the Earth's atmosphere is only on the order of parts per million, but H<sub>2</sub> is, in fact, the most abundant diatomic molecule in nature. The interstellar medium is, indeed, dominated by hydrogen atoms. | |||
The bond in a homonuclear diatomic molecule is non-polar. In most diatomic molecules, the elements are nonidentical. Prominent examples include [[carbon monoxide]], [[nitric oxide]], and [[hydrogen chloride]], but other important examples include gaseous MgO, SiO, and many other species not normally considered diatomic because they [[polymerize]] near room temperature. All halogens are diatomic, excepted [[astatine]]. | |||
Elements that consist of diatomic molecules, under typical laboratory conditions of 1 bar and 25 °C, include hydrogen (H<sub>2</sub>), nitrogen (N<sub>2</sub>), oxygen (O<sub>2</sub>), and the halogens (although it is not yet known whether [[astatine]] forms diatomic astatine molecules<ref>{{cite book|last=Hammond|first=C.R.|title=Handbook of Chemistry and Physics|year=2012|url=http://www.hbcpnetbase.com//articles/04_01_91.pdf|chapter=Section 4: Properties of the Elements and Inorganic Compounds}}</ref>).<ref>{{Cite book| author = Emsley, J. | title = The Elements | publisher = Oxford: Clarendon Press | year = 1989 | pages = 22–23| id= }}</ref> Other elements form homonuclear diatomics when evaporated, but these diatomic species repolymerize at lower temperatures. For example, heating ("cracking") elemental phosphorus gives [[diphosphorus]], P<sub>2</sub>. | |||
==Molecular geometry== | |||
{{Main|Molecular geometry}} | |||
Diatomic molecules cannot have any [[molecular geometry|geometry]] but linear, as any two points always lie in a line. This is the simplest [[stereochemistry|spatial arrangement of atoms]] after the sphericity of single atoms.<ref name = "qpxlgb">"VSEPR - A Summary". University of Berkeley College of Chemistry. 20 January 2008. http://mc2.cchem.berkeley.edu/VSEPR/</ref> | |||
==Historical significance== | |||
Diatomic elements played an important role in the elucidation of the concepts of element, atom, and molecule in the 19th century, because some of the most common elements, such as hydrogen, oxygen, and nitrogen, occur as diatomic molecules. [[John Dalton]]'s original atomic hypothesis assumed that all elements were monatomic and that the atoms in compounds would normally have the simplest atomic ratios with respect to one another. For example, Dalton assumed that water's formula was HO, giving the atomic weight of oxygen as eight times that of hydrogen, instead of the modern value of about 16. As a consequence, confusion existed regarding atomic weights and molecular formulas for about half a century. | |||
As early as 1805, [[Gay-Lussac]] and [[Alexander von Humboldt|von Humboldt]] showed that water is formed of two volumes of hydrogen and one volume of oxygen, and by 1811 [[Amedeo Avogadro]] had arrived at the correct interpretation of water's composition, based on what is now called [[Avogadro's law]] and the assumption of diatomic elemental molecules. However, these results were mostly ignored until 1860. Part of this rejection was due to the belief that atoms of one element would have no [[chemical affinity]] towards atoms of the same element, and part was due to apparent exceptions to Avogadro's law that were not explained until later in terms of dissociating molecules. | |||
At the 1860 [[Karlsruhe Congress]] on atomic weights, [[Cannizzaro]] resurrected Avogadro's ideas and used them to produce a consistent table of atomic weights, which mostly agree with modern values. These weights were an important pre-requisite for the discovery of the [[periodic law]] by [[Dmitri Mendeleev]] and [[Lothar Meyer]].<ref>{{Cite journal| author = Ihde, Aaron J. | title = The Karlsruhe Congress: A centennial retrospective | journal = Journal of Chemical Education | year = 1961 | volume = 38 | pages = 83–86 | url = http://search.jce.divched.org:8081/JCEIndex/FMPro?-db=jceindex.fp5&-lay=wwwform&combo=karlsruhe&-find=&-format=detail.html&-skip=0&-max=1&-token.2=0&-token.3=10 | accessdate=2007-08-24 | doi = 10.1021/ed038p83| issue = 2 |bibcode = 1961JChEd..38...83I }}</ref> | |||
==Excited electronic states== | |||
Diatomic molecules are normally in the lowest or ground state, which is also known as the <math>X</math> state. When a gas of diatomic molecules is bombarded by energetic electrons, the molecules are excited to higher electronic states, such as occurs, for example, in the natural aurora, high-altitude nuclear explosions, and rocket-born electron gun experiments.<ref name=gilmore1992/> The excitation can also occur when the gas absorbs light or other electromagnetic radiation. The excited states are unstable and naturally relax back to the ground state. Over various short time scales after the excitation (typically a fraction of a second, or sometimes longer than a second if the excited state is [[Metastability|metastable]]), transitions occur from the higher to lower electronic states and ultimately to the ground state, and each transition results in the emission of a photon. This emission is known as [[fluorescence]]. Successively higher electronic states are traditionally named <math>A</math>, <math>B</math>, <math>C</math>, etc. (but this convention is not always followed, and sometimes lower case letters and alphabetically out-of-sequence letters are used, as can be seen in the example given below). The excitation energy must be greater than or equal to the energy of the electronic state in order for the excitation to occur. | |||
In quantum theory, an electronic state of a diatomic molecule is represented by | |||
:<math>^{2S+1} \Lambda (v)</math> | |||
where <math>S</math> is the total electronic spin quantum number, <math>\Lambda</math> is the total electronic angular momentum quantum number along the internuclear axis, and <math>v</math> is the vibrational quantum number. <math>\Lambda</math> takes on values 0, 1, 2, …, which traditionally are represented by the electronic state symbols <math>\Sigma</math>, <math>\Pi</math>, <math>\Delta</math>,…. | |||
For example, the following table lists the common electronic states (without vibrational quantum numbers), along with the energy of the lowest vibrational level (<math>v=0</math>) of diatomic nitrogen (N<sub>2</sub>), the most abundant gas the the Earth's atmosphere.<ref name=laher1991/> In the table, the subscripts and superscripts after <math>\Lambda</math> give additional quantum mechanical details about the electronic state. | |||
{| class="wikitable" | |||
|- | |||
! State !! Energy (<math>T_0</math>, cm<math>^{-1}</math>) | |||
|- | |||
| <math>X ^1\Sigma_g^+</math> || 0.0 | |||
|- | |||
| <math>A ^3\Sigma_u^+</math> || 49754.8 | |||
|- | |||
| <math>B ^3\Pi_g</math> || 59306.8 | |||
|- | |||
| <math>W ^3\Delta_u</math> || 59380.2 | |||
|- | |||
| <math>B' ^3\Sigma_u^-</math> || 65851.3 | |||
|- | |||
| <math>a' ^1\Sigma_u^-</math> || 67739.3 | |||
|- | |||
| <math>a ^1\Pi_g</math> || 68951.2 | |||
|- | |||
| <math>w ^1\Delta_u</math> || 71698.4 | |||
|} | |||
==Energy levels== | |||
The [[molecular term symbol]] is a shorthand expression of the angular momenta that characterize the electronic quantum state of a diatomic molecule, which is an eigenstate of the electronic molecular [[Hamiltonian (quantum mechanics)|Hamiltonian]]. It is also convenient, and common, to represent a diatomic molecule as two-point masses connected by a massless spring. The energies involved in the various motions of the molecule can then be broken down into three categories: the translational, rotational, and vibrational energies. | |||
===Translational energies=== | |||
The translational energy of the molecule is simply given by the [[kinetic energy]] expression: | |||
:<math>E_{trans}=\frac{1}{2}mv^2</math> | |||
where ''m'' is the mass of the molecule and ''v'' is its velocity. | |||
===Rotational energies=== | |||
Classically, the kinetic energy of rotation is | |||
::<math>E_{rot} = \frac{L^2}{2 I} \,</math> | |||
:where | |||
::<math>L \,</math> is the [[angular momentum]] | |||
::<math>I \,</math> is the [[moment of inertia]] of the molecule | |||
For microscopic, atomic-level systems like a molecule, angular momentum can only have specific discrete values given by | |||
::<math>L^2 = l(l+1) \hbar^2 \,</math> | |||
:where ''l'' is a non-negative integer and <math>\hbar</math> is the [[reduced Planck constant]]. | |||
Also, for a diatomic molecule the moment of inertia is | |||
::<math>I = \mu r_{0}^2 \,</math> | |||
:where | |||
::<math>\mu \,</math> is the [[reduced mass]] of the molecule and | |||
::<math>r_{0} \,</math> is the average distance between the centers of the two atoms in the molecule. | |||
So, substituting the angular momentum and moment of inertia into E<sub>rot</sub>, the rotational energy levels of a diatomic molecule are given by: | |||
::<math style>E_{rot} = \frac{l(l+1) \hbar^2}{2 \mu r_{0}^2} \ \ \ \ \ l=0,1,2,... \,</math> | |||
===Vibrational energies=== | |||
Another way a diatomic molecule can move is to have each atom oscillate—or [[Vibration|vibrate]]—along a line (the bond) connecting the two atoms. The vibrational energy is approximately that of a [[quantum harmonic oscillator]]: | |||
::<math>E_{vib} = \left(n+\frac{1}{2} \right)\hbar \omega \ \ \ \ \ n=0,1,2,.... \,</math> | |||
:where | |||
::''n'' is an integer | |||
::<math>\hbar</math> is the [[reduced Planck constant]] and | |||
::<math>\omega</math> is the [[angular frequency]] of the vibration. | |||
===Comparison between rotational and vibrational energy spacings=== | |||
The spacing, and the energy of a typical spectroscopic transition, between vibrational energy levels is about 100 times greater than that of a typical transition between [[rotational energy]] levels. | |||
==Hund's cases== | |||
{{main|Hund's cases}} | |||
The [[good quantum number]]s for a diatomic molecule, as well as good approximations of rotational energy levels, can be obtained by modeling the molecule using [[Hund's cases]]. | |||
==Further reading== | |||
* {{Cite book| author = Huber, K. P. and Herzberg, G. | title = Molecular Spectra and Molecular Structure IV. Constants of Diatomic Molecules | publisher = New York: Van Nostrand: Reinhold | year = 1979 | id= }} | |||
* {{Cite book| author=Tipler, Paul | title=Physics For Scientists and Engineers : Vol. 1 (4th ed.) | publisher=W. H. Freeman | year=1998 | isbn=1-57259-491-8}} | |||
==See also== | |||
*[[AXE method]] | |||
*[[Octatomic element]] | |||
*[[Shared pair]] | |||
*[[Industrial gas]] | |||
==Notes and references== | |||
{{Reflist|2 | |||
<ref name=gilmore1992> | |||
{{cite journal | |||
| last = Gilmore | |||
| first = Forrest R. | |||
| display-authors = 3 | |||
| last2 = Laher | |||
| first2 = Russ R. | |||
| last3 = Espy | |||
| first3 = Patrick J. | |||
| year = 1992 | |||
| title = Franck-Condon Factors, r-Centroids, Electronic Transition Moments, and Einstein Coefficients for Many Nitrogen and Oxygen Band Systems | |||
| journal = [[Journal of Physical and Chemical Reference Data]] | |||
| volume = 21 | |||
| issue = 5 | |||
| pages = 1005-1107 | |||
}} | |||
</ref> | |||
<ref name=laher1991> | |||
{{cite journal | |||
| last = Laher | |||
| first = Russ R. | |||
| display-authors = 2 | |||
| last2 = Gilmore | |||
| first2 = Forrest R. | |||
| year = 1991 | |||
| title = Improved Fits for the Vibrational and Rotational Constants of Many States of Nitrogen and Oxygen | |||
| journal = [[Journal of Physical and Chemical Reference Data]] | |||
| volume = 20 | |||
| issue = 4 | |||
| pages = 685-712 | |||
}} | |||
</ref> | |||
}} | |||
==External links== | |||
* [http://hyperphysics.phy-astr.gsu.edu/hbase/molecule/rotrig.html#c3 Hyperphysics] – Rotational Spectra of Rigid Rotor Molecules | |||
* [http://hyperphysics.phy-astr.gsu.edu/hbase/quantum/hosc.html Hyperphysics] – Quantum Harmonic Oscillator | |||
* [http://www.3dchem.com/ 3D Chem] – Chemistry, Structures, and 3D Molecules | |||
* [http://www.iumsc.indiana.edu/ IUMSC] – Indiana University Molecular Structure Center | |||
{{MolecularGeometry}} | |||
{{diatomicelements}} | |||
{{DEFAULTSORT:Diatomic Molecule}} | |||
[[Category:Molecules]] | |||
[[Category:Stereochemistry]] | |||
[[Category:Molecular geometry]] | |||
Revision as of 21:04, 31 January 2014
30 year-old Entertainer or Range Artist Wesley from Drumheller, really loves vehicle, property developers properties for sale in singapore singapore and horse racing. Finds inspiration by traveling to Works of Antoni Gaudí.

Diatomic molecules are molecules composed of only two atoms, of either the same or different chemical elements. The prefix di- is of Greek origin, meaning "two". If a diatomic molecule consists of two atoms of the same element, such as hydrogen (H2) or oxygen (O2), then it is said to be homonuclear. Otherwise, if a diatomic molecule consists of two different atoms, such as carbon monoxide (CO) or boron monoxide (BO), the molecule is said to heteronuclear.
Homonuclear molecules
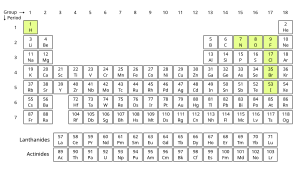
The only chemical elements which are stable two atom homonuclear molecules at standard temperature and pressure (STP), are hydrogen (H2), nitrogen (N2) and oxygen (O2), plus the halogens fluorine (F2) and chlorine (Cl2). Those diatomic elements that are gaseous at STP, when grouped together with the monatomic noble gases, such as argon, are called "elemental gases" or "molecular gases" to distinguish them from molecules that are also chemical compounds. The noble gases do not form diatomic molecules: this can be explained using molecular orbital theory.
The halogens bromine (Br2) and iodine (I2) can also form diatomic gas at slightly elevated temperatures.[1]
Other elements that can form two atom molecules are phosphorus (P2) and sulfur (S2) although neither of these molecules are stable in atmospheric conditions.
Heteronuclear molecules
All other diatomic molecules are chemical compounds of two different elements, for example, nitric oxide (NO). Many different elements combine to form heteronuclear diatomic molecules, and this phenomenon, in general, depends on temperature and pressure. Many chemical compounds form diatomic molecules when evaporated.
Occurrence
Hundreds of diatomic molecules have been characterized[2] in the terrestrial environment, laboratory, and interstellar medium. About 99% of the Earth's atmosphere is composed of two diatomic molecules: oxygen (21%) and nitrogen (78%). The natural abundance of hydrogen (H2) in the Earth's atmosphere is only on the order of parts per million, but H2 is, in fact, the most abundant diatomic molecule in nature. The interstellar medium is, indeed, dominated by hydrogen atoms.
The bond in a homonuclear diatomic molecule is non-polar. In most diatomic molecules, the elements are nonidentical. Prominent examples include carbon monoxide, nitric oxide, and hydrogen chloride, but other important examples include gaseous MgO, SiO, and many other species not normally considered diatomic because they polymerize near room temperature. All halogens are diatomic, excepted astatine.
Elements that consist of diatomic molecules, under typical laboratory conditions of 1 bar and 25 °C, include hydrogen (H2), nitrogen (N2), oxygen (O2), and the halogens (although it is not yet known whether astatine forms diatomic astatine molecules[3]).[4] Other elements form homonuclear diatomics when evaporated, but these diatomic species repolymerize at lower temperatures. For example, heating ("cracking") elemental phosphorus gives diphosphorus, P2.
Molecular geometry
Mining Engineer (Excluding Oil ) Truman from Alma, loves to spend time knotting, largest property developers in singapore developers in singapore and stamp collecting. Recently had a family visit to Urnes Stave Church.
Diatomic molecules cannot have any geometry but linear, as any two points always lie in a line. This is the simplest spatial arrangement of atoms after the sphericity of single atoms.[5]
Historical significance
Diatomic elements played an important role in the elucidation of the concepts of element, atom, and molecule in the 19th century, because some of the most common elements, such as hydrogen, oxygen, and nitrogen, occur as diatomic molecules. John Dalton's original atomic hypothesis assumed that all elements were monatomic and that the atoms in compounds would normally have the simplest atomic ratios with respect to one another. For example, Dalton assumed that water's formula was HO, giving the atomic weight of oxygen as eight times that of hydrogen, instead of the modern value of about 16. As a consequence, confusion existed regarding atomic weights and molecular formulas for about half a century.
As early as 1805, Gay-Lussac and von Humboldt showed that water is formed of two volumes of hydrogen and one volume of oxygen, and by 1811 Amedeo Avogadro had arrived at the correct interpretation of water's composition, based on what is now called Avogadro's law and the assumption of diatomic elemental molecules. However, these results were mostly ignored until 1860. Part of this rejection was due to the belief that atoms of one element would have no chemical affinity towards atoms of the same element, and part was due to apparent exceptions to Avogadro's law that were not explained until later in terms of dissociating molecules.
At the 1860 Karlsruhe Congress on atomic weights, Cannizzaro resurrected Avogadro's ideas and used them to produce a consistent table of atomic weights, which mostly agree with modern values. These weights were an important pre-requisite for the discovery of the periodic law by Dmitri Mendeleev and Lothar Meyer.[6]
Excited electronic states
Diatomic molecules are normally in the lowest or ground state, which is also known as the state. When a gas of diatomic molecules is bombarded by energetic electrons, the molecules are excited to higher electronic states, such as occurs, for example, in the natural aurora, high-altitude nuclear explosions, and rocket-born electron gun experiments.[7] The excitation can also occur when the gas absorbs light or other electromagnetic radiation. The excited states are unstable and naturally relax back to the ground state. Over various short time scales after the excitation (typically a fraction of a second, or sometimes longer than a second if the excited state is metastable), transitions occur from the higher to lower electronic states and ultimately to the ground state, and each transition results in the emission of a photon. This emission is known as fluorescence. Successively higher electronic states are traditionally named , , , etc. (but this convention is not always followed, and sometimes lower case letters and alphabetically out-of-sequence letters are used, as can be seen in the example given below). The excitation energy must be greater than or equal to the energy of the electronic state in order for the excitation to occur.
In quantum theory, an electronic state of a diatomic molecule is represented by
where is the total electronic spin quantum number, is the total electronic angular momentum quantum number along the internuclear axis, and is the vibrational quantum number. takes on values 0, 1, 2, …, which traditionally are represented by the electronic state symbols , , ,…. For example, the following table lists the common electronic states (without vibrational quantum numbers), along with the energy of the lowest vibrational level () of diatomic nitrogen (N2), the most abundant gas the the Earth's atmosphere.[8] In the table, the subscripts and superscripts after give additional quantum mechanical details about the electronic state.
| State | Energy (, cm) |
|---|---|
| 0.0 | |
| 49754.8 | |
| 59306.8 | |
| 59380.2 | |
| 65851.3 | |
| 67739.3 | |
| 68951.2 | |
| 71698.4 |
Energy levels
The molecular term symbol is a shorthand expression of the angular momenta that characterize the electronic quantum state of a diatomic molecule, which is an eigenstate of the electronic molecular Hamiltonian. It is also convenient, and common, to represent a diatomic molecule as two-point masses connected by a massless spring. The energies involved in the various motions of the molecule can then be broken down into three categories: the translational, rotational, and vibrational energies.
Translational energies
The translational energy of the molecule is simply given by the kinetic energy expression:
where m is the mass of the molecule and v is its velocity.
Rotational energies
Classically, the kinetic energy of rotation is
- where
- is the angular momentum
- is the moment of inertia of the molecule
For microscopic, atomic-level systems like a molecule, angular momentum can only have specific discrete values given by
- where l is a non-negative integer and is the reduced Planck constant.
Also, for a diatomic molecule the moment of inertia is
- where
- is the reduced mass of the molecule and
- is the average distance between the centers of the two atoms in the molecule.
So, substituting the angular momentum and moment of inertia into Erot, the rotational energy levels of a diatomic molecule are given by:
Vibrational energies
Another way a diatomic molecule can move is to have each atom oscillate—or vibrate—along a line (the bond) connecting the two atoms. The vibrational energy is approximately that of a quantum harmonic oscillator:
- where
- n is an integer
- is the reduced Planck constant and
- is the angular frequency of the vibration.
Comparison between rotational and vibrational energy spacings
The spacing, and the energy of a typical spectroscopic transition, between vibrational energy levels is about 100 times greater than that of a typical transition between rotational energy levels.
Hund's cases
Mining Engineer (Excluding Oil ) Truman from Alma, loves to spend time knotting, largest property developers in singapore developers in singapore and stamp collecting. Recently had a family visit to Urnes Stave Church. The good quantum numbers for a diatomic molecule, as well as good approximations of rotational energy levels, can be obtained by modeling the molecule using Hund's cases.
Further reading
- 20 year-old Real Estate Agent Rusty from Saint-Paul, has hobbies and interests which includes monopoly, property developers in singapore and poker. Will soon undertake a contiki trip that may include going to the Lower Valley of the Omo.
My blog: http://www.primaboinca.com/view_profile.php?userid=5889534 - 20 year-old Real Estate Agent Rusty from Saint-Paul, has hobbies and interests which includes monopoly, property developers in singapore and poker. Will soon undertake a contiki trip that may include going to the Lower Valley of the Omo.
My blog: http://www.primaboinca.com/view_profile.php?userid=5889534
See also
Notes and references
43 year old Petroleum Engineer Harry from Deep River, usually spends time with hobbies and interests like renting movies, property developers in singapore new condominium and vehicle racing. Constantly enjoys going to destinations like Camino Real de Tierra Adentro.
External links
- Hyperphysics – Rotational Spectra of Rigid Rotor Molecules
- Hyperphysics – Quantum Harmonic Oscillator
- 3D Chem – Chemistry, Structures, and 3D Molecules
- IUMSC – Indiana University Molecular Structure Center
Template:MolecularGeometry Template:Diatomicelements
- ↑ 20 year-old Real Estate Agent Rusty from Saint-Paul, has hobbies and interests which includes monopoly, property developers in singapore and poker. Will soon undertake a contiki trip that may include going to the Lower Valley of the Omo.
My blog: http://www.primaboinca.com/view_profile.php?userid=5889534 - ↑ 20 year-old Real Estate Agent Rusty from Saint-Paul, has hobbies and interests which includes monopoly, property developers in singapore and poker. Will soon undertake a contiki trip that may include going to the Lower Valley of the Omo.
My blog: http://www.primaboinca.com/view_profile.php?userid=5889534 - ↑ 20 year-old Real Estate Agent Rusty from Saint-Paul, has hobbies and interests which includes monopoly, property developers in singapore and poker. Will soon undertake a contiki trip that may include going to the Lower Valley of the Omo.
My blog: http://www.primaboinca.com/view_profile.php?userid=5889534 - ↑ 20 year-old Real Estate Agent Rusty from Saint-Paul, has hobbies and interests which includes monopoly, property developers in singapore and poker. Will soon undertake a contiki trip that may include going to the Lower Valley of the Omo.
My blog: http://www.primaboinca.com/view_profile.php?userid=5889534 - ↑ "VSEPR - A Summary". University of Berkeley College of Chemistry. 20 January 2008. http://mc2.cchem.berkeley.edu/VSEPR/
- ↑ One of the biggest reasons investing in a Singapore new launch is an effective things is as a result of it is doable to be lent massive quantities of money at very low interest rates that you should utilize to purchase it. Then, if property values continue to go up, then you'll get a really high return on funding (ROI). Simply make sure you purchase one of the higher properties, reminiscent of the ones at Fernvale the Riverbank or any Singapore landed property Get Earnings by means of Renting
In its statement, the singapore property listing - website link, government claimed that the majority citizens buying their first residence won't be hurt by the new measures. Some concessions can even be prolonged to chose teams of consumers, similar to married couples with a minimum of one Singaporean partner who are purchasing their second property so long as they intend to promote their first residential property. Lower the LTV limit on housing loans granted by monetary establishments regulated by MAS from 70% to 60% for property purchasers who are individuals with a number of outstanding housing loans on the time of the brand new housing purchase. Singapore Property Measures - 30 August 2010 The most popular seek for the number of bedrooms in Singapore is 4, followed by 2 and three. Lush Acres EC @ Sengkang
Discover out more about real estate funding in the area, together with info on international funding incentives and property possession. Many Singaporeans have been investing in property across the causeway in recent years, attracted by comparatively low prices. However, those who need to exit their investments quickly are likely to face significant challenges when trying to sell their property – and could finally be stuck with a property they can't sell. Career improvement programmes, in-house valuation, auctions and administrative help, venture advertising and marketing, skilled talks and traisning are continuously planned for the sales associates to help them obtain better outcomes for his or her shoppers while at Knight Frank Singapore. No change Present Rules
Extending the tax exemption would help. The exemption, which may be as a lot as $2 million per family, covers individuals who negotiate a principal reduction on their existing mortgage, sell their house short (i.e., for lower than the excellent loans), or take part in a foreclosure course of. An extension of theexemption would seem like a common-sense means to assist stabilize the housing market, but the political turmoil around the fiscal-cliff negotiations means widespread sense could not win out. Home Minority Chief Nancy Pelosi (D-Calif.) believes that the mortgage relief provision will be on the table during the grand-cut price talks, in response to communications director Nadeam Elshami. Buying or promoting of blue mild bulbs is unlawful.
A vendor's stamp duty has been launched on industrial property for the primary time, at rates ranging from 5 per cent to 15 per cent. The Authorities might be trying to reassure the market that they aren't in opposition to foreigners and PRs investing in Singapore's property market. They imposed these measures because of extenuating components available in the market." The sale of new dual-key EC models will even be restricted to multi-generational households only. The models have two separate entrances, permitting grandparents, for example, to dwell separately. The vendor's stamp obligation takes effect right this moment and applies to industrial property and plots which might be offered inside three years of the date of buy. JLL named Best Performing Property Brand for second year running
The data offered is for normal info purposes only and isn't supposed to be personalised investment or monetary advice. Motley Fool Singapore contributor Stanley Lim would not personal shares in any corporations talked about. Singapore private home costs increased by 1.eight% within the fourth quarter of 2012, up from 0.6% within the earlier quarter. Resale prices of government-built HDB residences which are usually bought by Singaporeans, elevated by 2.5%, quarter on quarter, the quickest acquire in five quarters. And industrial property, prices are actually double the levels of three years ago. No withholding tax in the event you sell your property. All your local information regarding vital HDB policies, condominium launches, land growth, commercial property and more
There are various methods to go about discovering the precise property. Some local newspapers (together with the Straits Instances ) have categorised property sections and many local property brokers have websites. Now there are some specifics to consider when buying a 'new launch' rental. Intended use of the unit Every sale begins with 10 p.c low cost for finish of season sale; changes to 20 % discount storewide; follows by additional reduction of fiftyand ends with last discount of 70 % or extra. Typically there is even a warehouse sale or transferring out sale with huge mark-down of costs for stock clearance. Deborah Regulation from Expat Realtor shares her property market update, plus prime rental residences and houses at the moment available to lease Esparina EC @ Sengkang - ↑ Cite error: Invalid
<ref>tag; no text was provided for refs namedgilmore1992 - ↑ Cite error: Invalid
<ref>tag; no text was provided for refs namedlaher1991

































